We use cookies to make your experience better. To comply with the new e-Privacy directive, we need to ask for your consent to set the cookies. Learn more.
 CUSTOM CAMELIDS VHH FRAGMENT
CUSTOM CAMELIDS VHH FRAGMENT
 About VHH
About VHH
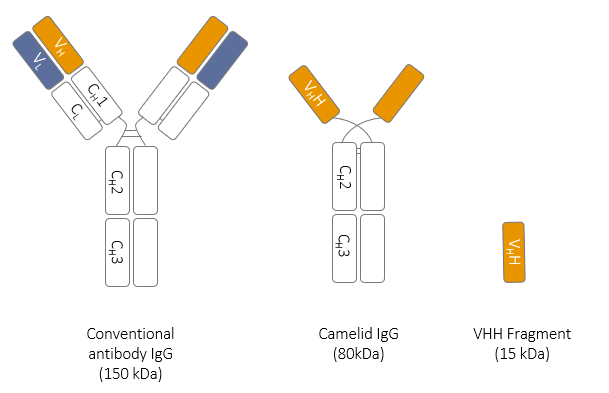
Camelids produce conventional antibodies made of 2 heavy chains and 2 light chains bound together with disulphide bonds in a Y shape. However, they also produce a unique subclasse of immunoglobulin G, also known as heavy chain IgG (hcIgG).
These antibodies are made of only two heavy chains without the CH1 region, and an antigen binding domain at their N-terminus called VHH.
The unique feature of hcIgG is the capacity of their monomeric antigen binding regions (VHH) to bind antigens with specificity, affinity and especially diversity that are comparable to conventional antibodies without the need of pairing with another region. Using molecular engeenering VHH fragaments can be generated.
 Why use VHH fragments ?
Why use VHH fragments ?
Bind the hidden epitope
Their steric hindrance is reduced, allowing them to reach hardly accessible antigens that conventional antibodies can not, such as active sites of enzymes (useful to inhibit enzyme activity) as well as smaller enzymes, such as lysozymes or ribonucleases.
Better permeability
Their capacity to penetrate tissues is increased compared to conventional antibodies, allowing a more precise staining of tissue sections when performing immunohistochemistry.
More stable
The high refolding capacity of the VHH region allow them to be thermodynamically more stable and resist to high temperatures (up to 70°C / 158°F) and extreme pH, such as gastric acid.
 VHH development
VHH development
1- CAMELID IMMUNIZATION
Immunization
According to our 88-day exclusive protocol, the immunization step consists in 4 injections and 2 test bleeds.
Monitoring
The immunoreactivity is monitored by the detection method developed for the project.
2- PHAGE LIBRARY CONSTRUCTION
PBMCs isolation and RNA extraction
Peripheral blood mononuclear cells (PBMCs) are isolated from the final bleed and total RNA is extracted.
cDNA synthesis and VHH amplification
RT-PCR and VHH antibody fragment amplification
Specific Primers are design to perform retro-transcription within the region of the coding for VHH and second nested PCR allows cDNA amplification.
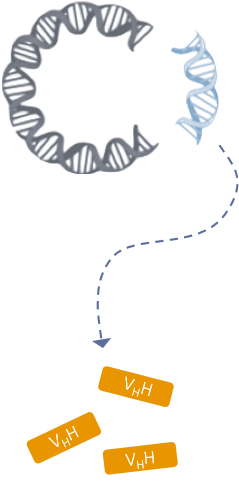
Phage vector cloning
VHH coding genes are integrated into a phage vector which is then used to transform competent bacteria for phage library production.

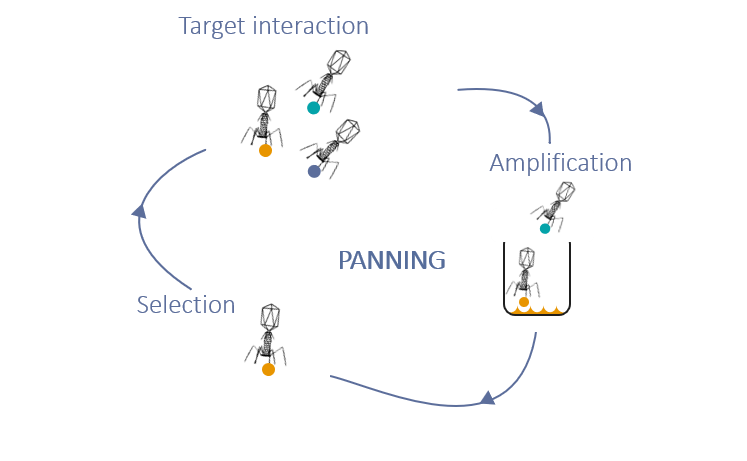
3- PANNING
Target interaction
The antigen is immobilized in the bottom of microtiter plate wells, phages preparations are loaded into the wells to allow them to bind the antigen through the VHH expressed at their surface.
Amplification
After washing off unbound phages, remaining phages are transduced into competent bacteria for enrichment and amplification.
Selection
At the end of each panning stage, cDNAs coding for VHH specific to the antigen are isolated
4- PRODUCTION AND PURIFICATION
Subcloning in an expression vector
Inserts are integrated into an expression vector, which is then used to transform competent bacteria.
Culture, lysis and affinity chromatography purification
Specific VHH are purified from culture supernatants by affinity chromatography.
All cDNAs coding for specific VHH are kept frozen in our laboratory.
 Testimonials
Testimonials
 Brochures
Brochures

Recombinant VHH
Discover our complete VHH development service.
